|
4/13/2024 0 Comments Molar mass periodic table trend The “key” that I am talking about is the Periodic Table of Elements. In fact, the use of this key is highly encouraged. This key is sort of like a secret key or “cheat” code in your favorite video game, that helps you successfully navigate to higher levels in the game. The difference is that the chemistry cheat code is actually something that is available in most science classrooms, textbooks, and online. It is available to everyone. 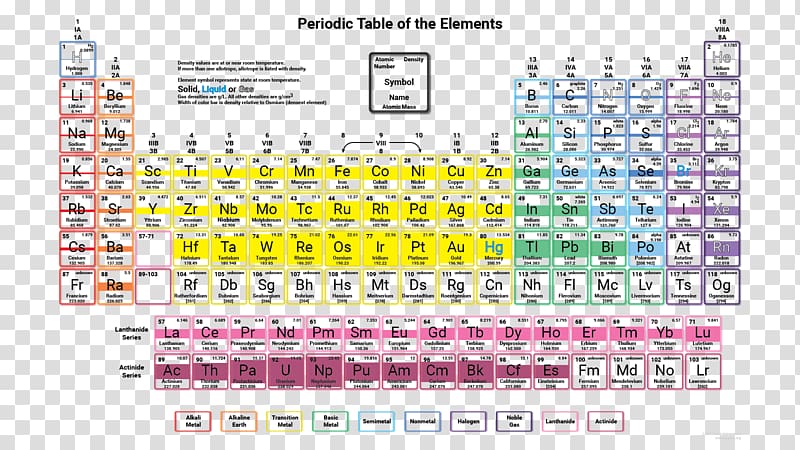
But what if I told you that there is a way that the memory requirements can be reduced a bit? What if I told you that there is actually a “key” that leads chemists to many (not all) of the answers that they might ask throughout their academic and professional careers? Would that be of some help? Even then, would that person have a thorough “understanding” of the material that was memorized? This is probably because of the perceived intricacies involved and all of the memory required to master the subject.Ĭertainly, if a person attempts to master the subject of chemistry through rote memorization the task would be quite arduous, if not insurmountable for all except those gifted with an eidetic memory. While this question may seem simple to answer, it actually demonstrates the use of one of the most basic, yet most important skills used in the subject and science of Chemistry.Ĭhemistry seems to be one of the most daunting and intimidating subjects for HighSchool and Undergrads to attempt to tackle. “ Which is the lightest element in the periodic table?” Please join me as we discuss how to answer one of the most elementary (pun intended) questions in the science of Chemistry. What will you do? Change your Major or program? Hire a tutor? Run away…? And atomic mass always increases in a period or a group.You have just learned that you have to take that most dreaded subject of all as part of your academic program. “CHEMISTRY.” 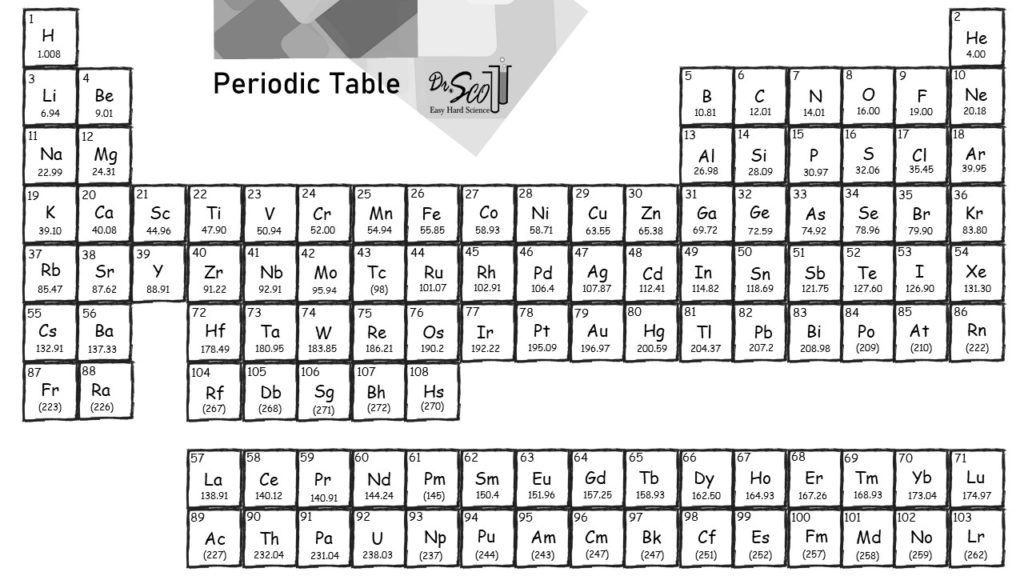
Note: Remember that the atomic masses which are mentioned in the periodic table of elements for different elements actually represent their average atomic masses. Thus, we can say that when we move down a group in case of non-metals, the reactivity of elements decreases while moving from left to right it increases in case of metals.Īlso, the atomic mass of the element actually uses average atomic masses of elements because naturally occurring elements exist as more than one isotopes which are the atoms of the same element which possess different relative masses. And the atomic mass on moving from left to right in the periodic table increases because the more protons and neutrons are added to the nuclei of an atom.Ītomic mass of an atom also increases while moving from top to bottom because the atoms in the rows, on further moving down, have more protons and nucleons. So, let us talk about one of the trends observed for the Atomic masses is defined as the sum of the total number of protons and neutrons of an atom. which increases or decreases down the group or while moving from left to bottom.Īs we know that a lot of trends are there in the physical and chemical properties of elements. Hint: As we know that the periodic table contains a trend for every parameter like atomic number, electronegativity, ionisation enthalpy, electron gain enthalpy etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
